Agile Stage-Gate: V-model + Agile for R&D projects
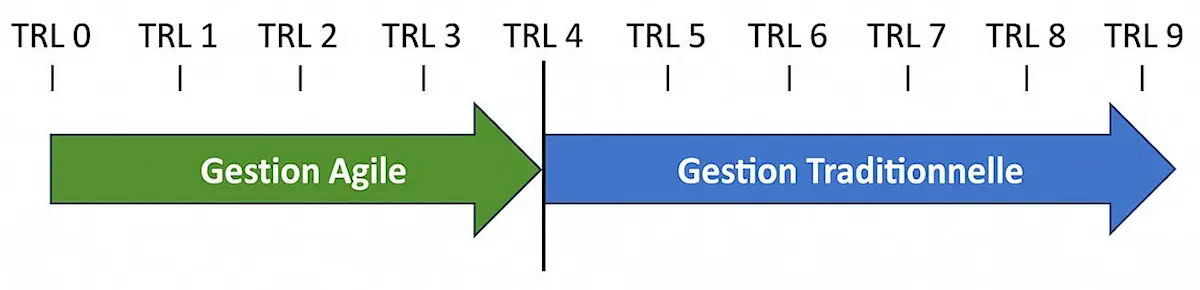
Hardware Agility does not replace the V-model, it complements it.
The exploration and technical validation phases benefit from short iterations to reduce risk. Industrialisation then follows a proven sequential process.
- Iterative validations in the early stages
- Controlled transition to industrialisation
- Compatible with ISO 9001, ISO 13485, GMP, DO-178C